Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Mesochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mesochem.com | |||
 | +86 (10) 5786-2036 57862181 67374028 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | sales@mesochem.com huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Apexbio Technology LLC | USA | |||
|---|---|---|---|---|
 | www.apexbt.com | |||
 | +1 (832) 696-8203 | |||
 | +1 (855) 527-3928 | |||
 | info@apexbt.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Liangjiang Medicine Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ljmedicine.com | |||
 | +86 (23) 6567-8473 | |||
 | +86 (23) 6567-8473 | |||
 | contact@ljmedicine.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Anhui Sun-wise Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunwisepharm.com | |||
 | +86 18856027890 | |||
 | sales@sunwisepharm.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jinan Create Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jngcpharm.com | |||
 | +86 (531) 8806-5293 | |||
 | +86 (531) 8806-5293 | |||
 | 3166282224@qq.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Antineoplastic agents >> Other antineoplastic agents |
|---|---|
| Name | Regorafenib monohydrate |
| Synonyms | 4-[4-[[[[4-Chloro-3-(trifluoromethyl)phenyl]amino]carbonyl]amino]-3-fluorophenoxy]-N-methyl-2-pyridinecarboxamide hydrate |
| Molecular Structure | 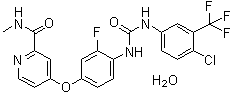 |
| Molecular Formula | C21H15ClF4N4O3.H2O |
| Molecular Weight | 500.83 |
| CAS Registry Number | 1019206-88-2 |
| EC Number | 851-928-5 |
| SMILES | CNC(=O)C1=NC=CC(=C1)OC2=CC(=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F)F.O |
| Solubility | Ethanol: 10 mM, DMSO: 40 mg/mL (Expl.) |
|---|---|
| Hazard Symbols |   GHS08;GHS09 Danger Details GHS08;GHS09 Danger Details |
|---|---|
| Risk Statements | H360Df-H360FD-H361fd-H362-H372-H373-H400-H410 Details |
| Safety Statements | P203-P260-P263-P264-P270-P273-P280-P318-P319-P391-P405-P501 Details |
| SDS | Available |
|
Regorafenib monohydrate is a small-molecule anticancer drug belonging to the class of multikinase inhibitors. It is used in oncology for the treatment of several advanced solid tumors. The compound exists as a monohydrate crystalline form, which is a solid-state variant containing one molecule of water associated with each molecule of regorafenib in the crystal lattice. This hydrated form is used in pharmaceutical formulations due to its stability and suitable physicochemical properties for oral administration. The active compound, regorafenib, was developed through medicinal chemistry efforts aimed at designing broad-spectrum inhibitors of protein kinases involved in tumor growth, angiogenesis, and the tumor microenvironment. It is structurally related to sorafenib, an earlier multikinase inhibitor, and was developed as part of a second-generation optimization strategy to improve potency and expand target coverage. Regorafenib was identified and developed by Bayer AG and advanced through preclinical and clinical evaluation for use in patients with refractory cancers. Regorafenib functions by inhibiting multiple receptor tyrosine kinases and serine/threonine kinases that regulate key biological processes in cancer progression. These include vascular endothelial growth factor receptors (VEGFRs), platelet-derived growth factor receptor (PDGFR), fibroblast growth factor receptor (FGFR), KIT, RET, and RAF kinases. By simultaneously targeting these signaling pathways, regorafenib interferes with tumor angiogenesis, tumor cell proliferation, and the survival of malignant cells. This broad inhibitory profile distinguishes it from more selective kinase inhibitors. The discovery of regorafenib was based on systematic structure–activity relationship studies of diaryl urea derivatives. Researchers modified the chemical scaffold of sorafenib to enhance binding affinity and broaden kinase inhibition profiles. These modifications included changes to the central phenyl-urea core and substitutions designed to improve pharmacokinetic properties and target engagement. The resulting compound demonstrated activity against a wider range of kinases while maintaining oral bioavailability. Regorafenib monohydrate is administered orally and undergoes absorption followed by extensive hepatic metabolism. The drug is primarily metabolized by cytochrome P450 enzymes, particularly CYP3A4, and undergoes further biotransformation into active metabolites that contribute to its pharmacological effects. Because of its metabolic profile, dosing must be carefully managed to balance efficacy and tolerability. Clinically, regorafenib is used for the treatment of metastatic colorectal cancer, gastrointestinal stromal tumors (GIST) that are resistant to imatinib and sunitinib, and hepatocellular carcinoma. It is typically administered in patients who have progressed on prior therapies. Its ability to inhibit angiogenic signaling is particularly important in these cancers, where tumor vascularization plays a central role in disease progression. The monohydrate form of regorafenib is important from a pharmaceutical development perspective because crystalline hydrates can influence drug stability, solubility, and manufacturing reproducibility. The presence of water in the crystal structure can affect dissolution behavior and shelf stability, making the monohydrate form suitable for consistent formulation into tablets. Regorafenib was approved for clinical use following large-scale clinical trials that demonstrated improvements in progression-free survival in patients with advanced cancers. Its introduction provided an additional therapeutic option for patients with limited treatment alternatives. However, its use is associated with a range of adverse effects typical of multikinase inhibitors, including hypertension, fatigue, hand–foot skin reaction, and gastrointestinal symptoms. These effects are related to its broad inhibition of signaling pathways in both tumor and normal tissues. Overall, regorafenib monohydrate is a broad-spectrum kinase inhibitor developed for the treatment of advanced solid tumors. Its discovery was based on rational modification of earlier kinase inhibitor scaffolds to enhance multi-target activity. The compound acts by inhibiting key signaling pathways involved in tumor growth and angiogenesis, and its monohydrate crystalline form supports its pharmaceutical use in oral therapy. References 2025. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2024 for the treatment of colorectal cancer. International Journal of Clinical Oncology. DOI: 10.1007/s10147-025-02899-8 2022. Development and validation of a simple and sensitive HPLC method for the determination of related substances in regorafenib tablets. Analytical sciences : the international journal of the Japan Society for Analytical Chemistry. DOI: 10.1007/s44211-022-00068-9 2016. Comparison of the crystal structures of the potent anticancer and anti-angiogenic agent regorafenib and its monohydrate. Acta crystallographica. Section C, Structural chemistry. DOI: 10.1107/s2053229616003727 |
| Market Analysis Reports |