Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Alsachim SAS | France | |||
|---|---|---|---|---|
 | www.alsachim.com | |||
 | +33 (0) 390 402 200 | |||
 | +33 (0) 390 402 199 | |||
 | contact@alsachim.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Opicapone |
| Synonyms | 5-[3-(2,5-Dichloro-4,6-dimethyl-1-oxido-3-pyridinyl)-1,2,4-oxadiazol-5-yl]-3-nitro-1,2-benzenediol; BIA 9-1067 |
| Molecular Structure | 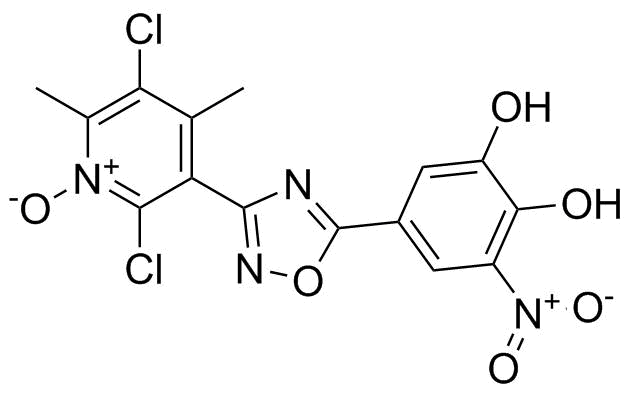 |
| Molecular Formula | C15H10Cl2N4O6 |
| Molecular Weight | 413.17 |
| CAS Registry Number | 923287-50-7 |
| EC Number | 810-046-0 |
| SMILES | CC1=C(C(=[N+](C(=C1Cl)C)[O-])Cl)C2=NOC(=N2)C3=CC(=C(C(=C3)O)O)[N+](=O)[O-] |
| Solubility | Practically insoluble (0.038 g/L) (25 $degree$C), Calc.* |
|---|---|
| Density | 1.80$+/-$0.1 g/cm3 (20 $degree$C 760 Torr), Calc.*78 |
| Boiling point | 701.1$+/-$70.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 377.8$+/-$35.7 $degree$C (Calc.)* |
| Index of refraction | 1.741 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS02 DangerGHS02; Details GHS02 DangerGHS02; Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228 Details | ||||||||||||
| Safety Statements | P210-P240-P241-P280-P370+P378 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Opicapone is a synthetic small-molecule drug used as an adjunct treatment in Parkinson’s disease. It is a potent and selective inhibitor of catechol-O-methyltransferase (COMT), an enzyme involved in the metabolic breakdown of levodopa, the primary therapeutic agent used to manage Parkinsonian symptoms. Chemically, opicapone is a substituted benzoic acid derivative containing a nitrobenzoxadiazole-related heteroaromatic system and a pyridine-like moiety within a highly conjugated structure. The molecule was designed to achieve strong, long-lasting binding to COMT, with a pharmacokinetic profile suitable for once-daily oral administration. The mechanism of action of opicapone is based on inhibition of peripheral COMT. COMT catalyzes the methylation of levodopa to 3-O-methyldopa, a metabolite that reduces the availability of levodopa for transport into the brain. By inhibiting COMT in peripheral tissues, opicapone decreases levodopa metabolism, thereby increasing its plasma half-life and improving its delivery to the central nervous system. This results in enhanced and more sustained dopaminergic stimulation in the brain when used in combination with levodopa/carbidopa therapy. Opicapone acts as a reversible, long-acting inhibitor that binds to COMT with high affinity. Unlike earlier COMT inhibitors such as entacapone, opicapone has a prolonged duration of action, allowing once-daily dosing. Its binding is characterized by slow dissociation from the enzyme, contributing to sustained inhibition even after plasma concentrations decline. The development of opicapone was driven by the need to improve motor fluctuations in Parkinson’s disease patients receiving levodopa therapy. As Parkinson’s disease progresses, patients often experience “wearing-off” phenomena due to fluctuating levodopa levels. COMT inhibition helps stabilize these levels and extend the duration of therapeutic effect. After oral administration, opicapone is absorbed and undergoes hepatic metabolism. It is primarily converted to inactive metabolites through reduction and conjugation pathways. The parent compound exhibits limited systemic exposure relative to its pharmacodynamic effect due to tight enzyme binding and slow dissociation. Clinically, opicapone is used as an add-on therapy to levodopa/carbidopa in adult patients with Parkinson’s disease experiencing end-of-dose motor fluctuations. It is not used as monotherapy and is intended to prolong the effect of each levodopa dose rather than replace dopaminergic therapy. Common adverse effects associated with opicapone include dyskinesia (often due to increased levodopa exposure), insomnia, dizziness, constipation, and orthostatic hypotension. These effects are generally consistent with enhanced dopaminergic activity. From a pharmacological perspective, opicapone represents a third-generation COMT inhibitor with improved potency, selectivity, and dosing convenience compared with earlier agents. Its design focuses on peripheral COMT inhibition, minimizing central nervous system penetration while maximizing systemic levodopa availability. Overall, opicapone is a once-daily, long-acting COMT inhibitor used in Parkinson’s disease to enhance and prolong the effect of levodopa therapy. By blocking peripheral levodopa metabolism, it increases and stabilizes dopaminergic signaling in the brain, helping reduce motor fluctuations associated with disease progression and chronic treatment. References 2026. Pharmacokinetics and pharmacodynamics of anti-Parkinson drugs in geriatric patients : Key for optimization of treatment. Zeitschrift fur Gerontologie und Geriatrie. URL: https://pubmed.ncbi.nlm.nih.gov/41711898 2026. Advancing the Treatment of Motor Fluctuations in Parkinson’s Disease with a Next-Generation Levodopa/Carbidopa Formulation. Neurology and Therapy. URL: https://pubmed.ncbi.nlm.nih.gov/41724884 2025. Safety of COMT-inhibitors in parkinson’s disease: a phase-IV comparative study on adverse events of Tolcapone, Entacapone and Opicapone. Journal of neural transmission (Vienna, Austria : 1996). URL: https://pubmed.ncbi.nlm.nih.gov/41175235 |
| Market Analysis Reports |